Host Cell Protein (HCP) Analysis Service
Many biotherapeutical products, including antibodies, vaccines, and recombinant proteins, are generated through biological processes. Despite of extensive purification steps employed, residual contaminants, as called host cell proteins (HCP), from the biological systems that are used to generate the biologics may still present in the final product. These HCPs, even of low levels, may compromise the safety and efficacy of the biotherapeutical products. Thus, detection and evaluation of HCP in biotherapeutical products are essential for quality and safety control.
MtoZ Biolabs uses 2D DIGE and Western blot technology for HCP analysis. This technology offers high sensitivity and can be employed for analyzing HCP from multiple species, such as mammalian cells, yeast, bacteria, and so on. Just tell us the potential HCP that may be contained in your sample, we will take charge of all the following analysis.
Analysis Workflow
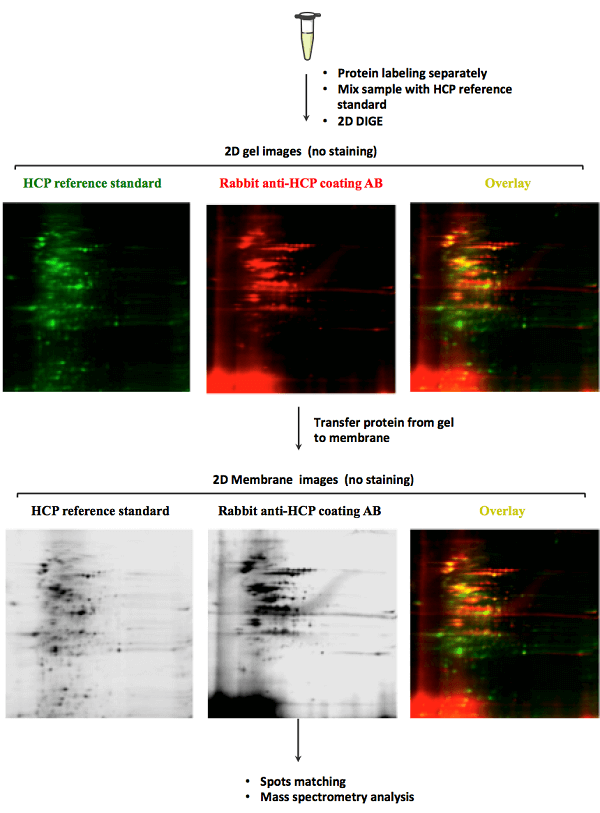
Service Advantages
1. Characterize HCP Originated from Multiple Species
2. Comprehensive and Accurate Evaluation of Residual HCP
3. Wide Dynamic Range and High Sensitivity, Enabling Detection of Trace Amount of HCP Contaminants
4. Highly Efficient Analysis and Short Turnaround Time
Applications
1. Characterizations of HCP in Antibody Products
2. Detection of HCP Contaminants in Recombinant Proteins
3. Detection of HCP Contaminants in Therapeutic Proteins and Vaccines
Deliverables
1. Information of Experimental Procedures and Reagents Used
2. Parameters of 2D DIGE System and Mass Spectrometry
3. HCP Identification
4. Raw Data Files
Related Services
Identification of Biopharm
Variation Analysis
Purity Analysis
• Host Cell Protein (HCP) Content Detection Service
Host cell proteins (HCP) content analysis, a critical quality control measure, is essential for determining and quantifying the content of HCP in the final product during bioproduct production. Primary techniques for assessing HCP content include immunological methods, notably enzyme-linked immunosorbent assay (ELISA), and mass spectrometry (MS). ELISA can roughly evaluate HCP levels by detecting HCP-specific antigens. Conversely, liquid chromatography-tandem mass spectrometry (LC-MS/MS) provides deta......
• HCP Identification and Relative Quantitative Analysis Service
Host cell proteins (HCP) are proteins expressed by production cells and may remain in the final product during bioproduct manufacturing. HCP is a critical quality marker in the development and production of bioproducts, particularly recombinant protein drugs. Its presence can impact drug purity and stability and may trigger adverse immune responses. Thus, qualitative identification and relative quantification of HCP are essential in drug development and quality control.
• HCP Absolute Quantification Analysis Service
Host Cell Proteins (HCP) are a very important class of impurities in the biopharmaceutical process. When host cells such as E. coli, yeast, mammalian cells, etc., are used to produce recombinant proteins or other biologics, their own proteins may mix with the target product. These residual HCPs can not only affect the safety and efficacy of drugs but may also provoke adverse reactions or immune responses.
• Host Cell Protein (HCP) Antibody Coverage Analysis Service
HCP antibody coverage refers to the proportion of specific host cell proteins (HCPs) detected by antibodies during pharmaceutical production. HCPs are protein impurities that inevitably remain in the final product during the production of recombinant proteins or monoclonal antibodies using host cells (such as E. coli, mammalian cells, etc.). These HCPs may affect the safety and efficacy of the drug, and therefore must be removed or reduced to acceptable levels through specific purification steps.
How to order?



